Selecting the wrong battery electrodes leads to premature failure and costly product recalls. Engineers often struggle to balance energy density and cycle life. We provide clear guidelines to help you optimize battery designs by understanding electrode fundamentals and material interactions.
Anode vs cathode refers to the two essential electrodes in a battery. The anode meaning relates to the electrode where oxidation occurs, releasing electrons. The cathode is where reduction happens, absorbing electrons. Together, these components determine the voltage, capacity, and lifespan of a cell.
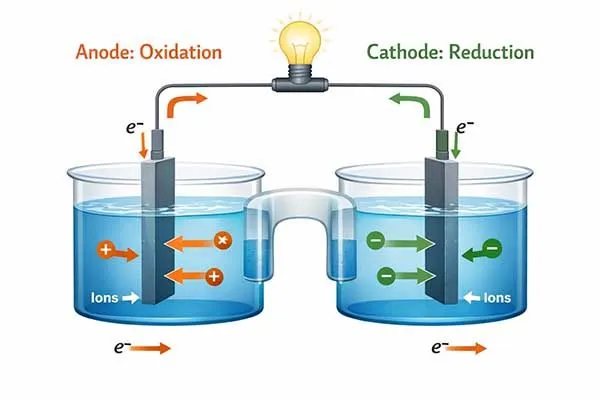
Understanding the precise roles of the anode vs cathode in lithium ion battery systems is critical for engineers designing systems for electric mobility, energy storage, and portable devices.
Let us examine the specific electrochemical mechanisms, material choices, and manufacturing processes that dictate battery performance in modern engineering applications.
Table of Contents
- What is an Anode and Cathode?
- How Do Anode and Cathode Roles Change in Rechargeable Batteries?
- What Are the Electrode Material Choices and Why Do They Matter?
- How Do Manufacturing and Functional Impacts Alter Battery Design?
- What Are the Practical Implications for EV, ESS & Portable Applications?
What is an Anode and Cathode?
The anode and cathode represent the negative and positive terminals during discharge. The anode releases electrons through oxidation, while the cathode accepts them via reduction. This fundamental chemical reaction powers external circuits.
Grasping the true anode meaning ensures engineers can properly model the electron flow and electrochemical potential[1] when developing new cell architectures for various power applications.
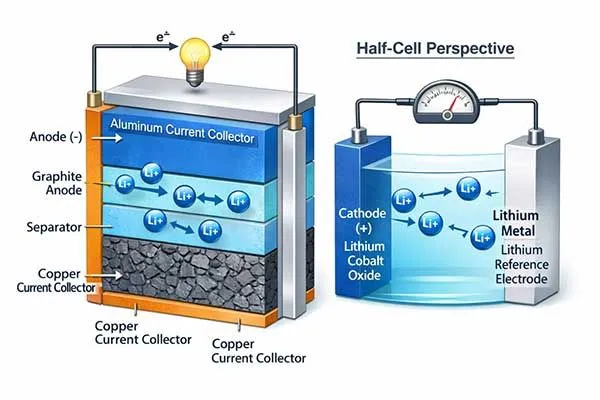
Exploring the Core Meaning and Electrochemical Pathways
To truly comprehend battery mechanics, engineers must start with simple terms before moving to precise definitions. In layman’s terms, people often consider the anode as the “minus” side and the cathode as the “plus” side of a battery.
However, from a strict electrochemical standpoint, the anode meaning relies entirely on the flow of electrons. The anode is the electrode where oxidation takes place. During oxidation, the electrode loses electrons. Conversely, the cathode is the electrode where reduction occurs, meaning it gains electrons.
Tips
In lithium-based systems, electrode balance directly impacts cycle life and safety margins. Always evaluate anode-to-cathode capacity ratio during early-stage battery design.
When analyzing anode vs cathode interactions, we must view the battery as a complete circuit. During a standard discharge cycle, lithium ions travel through the electrolyte and separator[2] from the anode to the cathode. Simultaneously, the liberated electrons cannot pass through the electrically insulating separator.
Instead, they flow through the external circuit, providing the electrical current needed to power devices ranging from power tools to unmanned aerial vehicles. We must evaluate anode vs cathode dynamics critically; one cannot function without the other.
If the anode releases electrons faster than the cathode can accept them, internal resistance spikes, leading to heat generation.
| Electrode Component | Chemical Process | Electron Action | Ion Movement (Discharge) |
|---|---|---|---|
| Anode (Negative Terminal) | Oxidation | Releases Electrons | Releases Li-ions into electrolyte |
| Cathode (Positive Terminal) | Reduction | Accepts Electrons | Absorbs Li-ions from electrolyte |
A dialectical approach to designing these systems requires engineers to question the theoretical limits of these materials against their practical limitations. For instance, while a specific material might offer an excellent electrochemical reaction path for rapid oxidation, its structural integrity might degrade if it expands too much during the process.
Therefore, evaluating anode vs cathode performance is never just about voltage; it is about finding an equilibrium between electron release rates, ion diffusion speeds, and structural stability over thousands of cycles.
Master Anode & Cathode Fundamentals
Optimize your battery designs today.
How Do Anode and Cathode Roles Change in Rechargeable Batteries?
In rechargeable systems, the anode and cathode reverse roles during charging. The discharge anode becomes the charge cathode, and vice versa.
However, any standard lithium ion battery manufacturer conventionally names the electrodes based on their discharge states to avoid engineering confusion. This means the negative electrode always retains the title of anode in industry documentation.
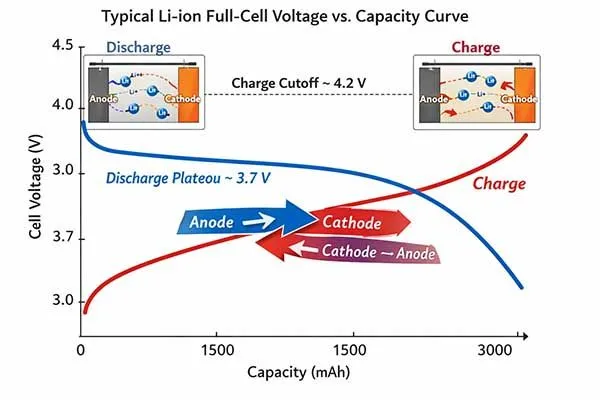
The Dynamic Shift: Discharge vs Charge Roles
The terminology surrounding rechargeable batteries often creates confusion because the electrochemical roles of the electrodes are not static. To fully understand anode vs cathode behavior in secondary (rechargeable) batteries, engineers must critically examine the discharge vs charge roles[3].
When you connect a battery to a load, the cell discharges. In this state, the negative electrode oxidizes and acts as the anode, while the positive electrode reduces and acts as the cathode.
However, when you plug the battery into a charger, you force current into the cell in the reverse direction. This reversal forces the positive electrode to undergo oxidation (releasing electrons and lithium ions) and the negative electrode to undergo reduction (absorbing electrons and lithium ions).
Therefore, during the charging phase, the physical positive terminal temporarily adopts the true electrochemical anode meaning, and the negative terminal becomes the cathode.
| Battery State | Negative Electrode acts as… | Positive Electrode acts as… | Li-ion Flow Direction |
|---|---|---|---|
| Discharging (Providing Power) | Anode (Oxidation) | Cathode (Reduction) | Negative to Positive |
| Charging (Receiving Power) | Cathode (Reduction) | Anode (Oxidation) | Positive to Negative |
Despite this dynamic electrochemical shift, changing the names of the physical components based on their state of charge would cause immense confusion in manufacturing and assembly. To solve this, the battery industry universally standardizes the terms based solely on the discharge state.
We evaluate the anode vs cathode relationship knowing that the physical materials remain fixed, even as their chemical roles invert. This dialectical contradiction—static naming versus dynamic chemical reality—requires engineers to maintain a dual perspective.
When designing charge profiles or calculating thermal loads during fast charging, you must remember that the “anode” is acting as a cathode. This understanding allows for precise tuning of charging algorithms[4] to prevent lithium plating[5], a dangerous condition where lithium ions accumulate on the anode surface instead of safely intercalating into the structure during rapid charge cycles.
What Are the Electrode Material Choices and Why Do They Matter?
Material selection dictates battery capabilities entirely. A detailed battery anode vs cathode material comparison reveals trade-offs between cost, safety, and energy density.
Common anodes use graphite or silicon, while cathodes utilize LFP or NMC compounds. Choosing the correct anode vs cathode pairing ensures the battery meets strict application requirements without compromising operational safety.

Evaluating Mainstream Electrode Materials
The selection of active materials is the most critical decision a lithium ion battery manufacturer makes. When we conduct a graphite vs silicon anode comparison, we see a classic engineering trade-off.
Graphite[6] remains the industry standard due to its excellent structural stability, low material cost, and reliable supply chain. It provides a stable cycle life because it expands very little (around 10%) when absorbing lithium ions. However, its theoretical capacity is limited to 372 mAh/g.
Silicon[7], on the other hand, offers an incredibly high theoretical capacity (nearly 3,600 mAh/g), but it suffers from massive volume expansion (up to 300%) during lithiation.
This expansion pulverizes the electrode and destroys the battery’s cycle life. Critical thinking dictates that instead of a pure silicon anode, engineers blend small percentages of silicon oxide into graphite to boost capacity while maintaining mechanical integrity.
| Material Type | Common Examples | Key Advantages | Primary Trade-offs |
|---|---|---|---|
| Anode | Graphite | High stability, low cost, excellent cycle life | Lower theoretical capacity limit |
| Anode | Silicon (Blended) | Significantly higher energy capacity | Massive volume expansion, reduced cycle life |
| Cathode | LFP (LiFePO4) | High safety, long cycle life, low cost, cobalt-free | Lower operating voltage, lower energy density |
| Cathode | NMC (LiNiMnCoO2) | High energy density, excellent power output | Higher cost, thermal runaway risks, supply chain issues |
The cathode side presents an equally complex dialectic. An LFP cathode vs NMC cathode analysis highlights the tension between safety and energy density.
Lithium Iron Phosphate (LFP) offers exceptional thermal stability, a longer cycle life, and utilizes highly abundant, lower-cost materials (iron and phosphorus), free from controversial metals like cobalt. LFP is incredibly safe, making it ideal for energy storage systems and entry-level electric mobility. However, its energy density usually maxes out around 160 Wh/kg.
NMC (Nickel Manganese Cobalt) cathodes offer much higher energy density, often exceeding 250 Wh/kg, making them the preferred choice for long-range electric vehicles. The downside is a higher material cost, a more vulnerable supply chain, and lower thermal runaway thresholds.
| Material | Typical Capacity (mAh/g) | Voltage vs Li | Key Advantage | Common Use |
|---|---|---|---|---|
| Graphite | 372 | ~0.1 V | Long cycle life | EV, ESS |
| Silicon blend | 600–1200 | ~0.2 V | High energy density | Premium EV |
| LFP | 160 | 3.2 V | Safety, long life | ESS, EV |
| NMC | 180–220 | 3.6–3.7 V | High energy density | EV |
| Li-SOCl₂ | — | 3.6 V | Ultra long shelf life | Metering, IoT |
When analyzing anode vs cathode interactions, one must remember that the overall cell voltage and capacity are limited by the weakest link. You cannot pair a massive capacity anode with a low-voltage cathode and expect revolutionary results. The anode vs cathode matching process requires precise capacity balancing.
Choose Optimal Anode/Cathode Chemistries
Balance energy, cost & safety.
How Do Manufacturing and Functional Impacts Alter Battery Design?
Manufacturing processes directly dictate how anode affects battery performance and how cathode affects energy density. Precise electrode coating[8], calendering, and material ratio adjustments determine the final cell specifications.
Optimizing these complex manufacturing steps is crucial for balancing the battery weight vs energy density and extending the overall cycle life of the pack.
Engineering the Balance: From Slurry to Final Cell
Theoretical electrochemistry only works if the manufacturing process is flawless. The journey from raw powder to a finished cylindrical battery or prismatic battery involves meticulous control. The process begins with mixing the active materials, binders, and conductive additives into a uniform slurry.
This slurry is then coated onto metallic foils—copper for the anode and aluminum for the cathode. The drying and calendering[9] (pressing) stages define the porosity and density of the electrodes. If you compress the electrode too much to increase energy density, you restrict the ion pathways, which severely degrades fast-charging capabilities.
| Manufacturing Stage | Process Description | Impact on Anode vs Cathode Performance |
|---|---|---|
| Mixing & Slurry Preparation | Blending active materials with binders and solvents. | Ensures uniform electron distribution and prevents localized hot spots. |
| Coating & Drying | Applying slurry to Copper (Anode) and Aluminum (Cathode) foils. | Determines mass loading, directly affecting total cell capacity. |
| Calendering | Compressing the coated foils via heavy rollers. | Balances energy density (thickness) against ion mobility (porosity). |
Tips
Material purity and coating uniformity are often more critical than nominal chemistry when targeting long-term field reliability.
Our Sales Manager Luke Liu recently engaged a US client requiring a specialized light electric vehicle battery that demanded both exceptional capacity and longevity. They struggled to find a manufacturer capable of balancing these conflicting metrics.
Our Chief Engineer Jack Song led a dedicated prototyping phase at Long Sing Energy. We approached the problem critically, knowing that the electrode material impact on cycle life is heavily influenced by the N/P (Negative-to-Positive) capacity ratio.
During our prototyping, we found that simply using a high-nickel NMC cathode caused premature degradation due to internal stress. We solved the problem through meticulous raw material matching and specific electrode ratio adjustments. By fine-tuning the N/P ratio[10] to ensure the anode had just enough excess capacity to prevent lithium plating without adding dead weight, we optimized the battery weight vs energy density.
Furthermore, we redesigned the thermal management design[11] to dissipate heat evenly across the cylindrical cells. This holistic view of anode vs cathode engineering allowed us to deliver a product that met the US client’s rigorous demands. Designing the ideal anode vs cathode architecture requires a constant dialectical negotiation between the chemical ideal and the manufacturing reality.
Need Reliable Battery Manufacturing Support?
Early collaboration with battery manufacturers can significantly reduce redesign cycles in custom pack development. Scale with proven production.
What Are the Practical Implications for EV, ESS & Portable Applications?
Field applications require strict adherence to a battery electrode material selection guide. Electric vehicles prioritize maximum energy density, while energy storage systems emphasize safety, cycle life, and sustainability.
Selecting the correct anode vs cathode combination ensures optimal performance, thermal stability, and long-term viability across diverse and demanding industrial use cases.

Aligning Material Properties with Application Demands
When an engineer drafts a specification sheet, the intended application dictates every anode vs cathode decision. We must adopt a critical mindset when evaluating the practical implications of our designs.
There is no universally perfect battery; there are only optimized compromises. For electric mobility and light electric vehicles, range anxiety is the primary consumer pain point. Therefore, manufacturers lean heavily toward NMC cathodes paired with graphite-silicon composite anodes to maximize energy density.
However, this anode vs cathode configuration requires robust, liquid-cooled thermal management systems[12] to mitigate safety risks associated with high-nickel chemistry.
Conversely, Energy Storage Systems (ESS) operate under entirely different constraints. Weight and volume are secondary concerns compared to operational lifespan and safety.
In this sector, the LFP cathode paired with a standard graphite anode is the dominant choice. This anode and cathode combination provides exceptional cycle life, often exceeding 6,000 cycles, and offers inherent thermal stability, drastically reducing the risk of catastrophic fires in large-scale grid installations.
Furthermore, as the industry moves toward greener practices, we must emphasize recyclable materials[13] and sustainability. LFP batteries are easier to recycle and do not rely on ethically complex cobalt supply chains, aligning well with global sustainability goals.
| Application | Primary Requirement | Optimal Anode vs Cathode Choice |
|---|---|---|
| Electric Vehicles (EV) | High Energy Density, Long Range | Silicon-Graphite Anode / High-Nickel NMC Cathode |
| Energy Storage Systems (ESS) | Long Cycle Life, Safety, Low Cost | Graphite Anode / LFP Cathode |
| Power Tools & UAVs | High Power Output (High C-Rate) | Graphite Anode / NMC or NCA Cathode (Thinly Coated) |
When developing battery packs for unmanned aerial vehicles (UAVs) or high-drain power tools, the anode vs cathode dynamics shift toward discharge rates. These applications demand massive bursts of power. Engineers must design thin electrodes with high porosity to facilitate rapid ion transfer, deliberately sacrificing some overall capacity to achieve high C-rates[14].
Understanding the deeper anode meaning—that it is the source of the electron flood—reminds us that the anode structure must rapidly release ions without physically degrading under extreme load. By critically analyzing the specific needs of each sector, engineers can tailor the anode and cathode pairings to deliver safe, reliable, and sustainable energy solutions.
Looking for Application-Specific Battery Solutions?
Conclusion
Mastering the fundamental concepts of anode vs cathode electrochemistry allows engineers to design superior energy solutions. By carefully selecting active materials like LFP, NMC, graphite, and silicon, manufacturers can successfully navigate the engineering trade-offs between energy density, safety, and cycle life.
Optimizing manufacturing processes and maintaining strict quality control ensures that the final battery packs meet the demanding requirements of modern electric mobility, energy storage systems, and specialized industrial equipment while pushing toward a more sustainable future.
Frequently Asked Questions
Click to explore more information about Anode vs Cathode
Q: What is the difference between an anode and a cathode?
A: An anode is the electrode where oxidation occurs (loss of electrons), while a cathode is where reduction occurs (gain of electrons). In a discharging battery (galvanic cell), the anode is the negative electrode, and the cathode is the positive electrode. Roles reverse during charging in rechargeable cells.
Q: What is the difference between cathode, anode, and cation, and anion?
A: Cathode = electrode where reduction (gain of electrons) happens. Anode = electrode where oxidation (loss of electrons) happens. Cation = positively charged ion (attracted to the cathode, the negative electrode in electrolytic cells). Anion = negatively charged ion (attracted to the anode, the positive electrode in electrolytic cells). Cations move toward the cathode; anions move toward the anode.
Q: Is cathode always positive or negative?
A: No, the cathode is not always positive or negative. It is defined by the reaction: reduction occurs at the cathode. In discharging batteries (galvanic/voltaic cells), the cathode is positive. In charging batteries or electrolytic cells, the cathode is negative.
Q: Why are (+) and (-) signs marked on a cell?
A: The (+) and (-) signs indicate the polarity during discharge: (+) marks the positive terminal (cathode in discharge, where electrons enter the external circuit), and (-) marks the negative terminal (anode in discharge, where electrons exit to the external circuit). This helps users connect devices correctly for power delivery.
Q: Is the anode on the left or right?
A: There is no universal rule—convention varies by diagram. In many textbook galvanic cell diagrams (e.g., Daniell cell), the anode (negative, oxidation) is shown on the left and the cathode (positive, reduction) on the right, but placement depends on the illustration and is not fixed.
Q: What is the purpose of an anode?
A: The anode’s purpose is to undergo oxidation, releasing electrons into the external circuit (in batteries) or accepting electrons from species in solution (in electrolysis). It acts as the source of electrons in galvanic cells, driving current flow.
Q: Where are cathodes used?
A: Cathodes are used in batteries (e.g., Li-ion, alkaline), electrolytic cells (e.g., electroplating, aluminum production), fuel cells, corrosion protection (sacrificial cathodes), vacuum tubes (CRT), and medical devices (e.g., defibrillators). They are the site of reduction reactions.
Q: Why do electrons flow from anode to cathode?
A: Electrons flow from anode to cathode in the external circuit because oxidation at the anode generates free electrons (making it the negative source), while reduction at the cathode consumes electrons (making it the positive sink). This creates a spontaneous electron flow from higher to lower potential in galvanic cells.
Q: Is lithium a cathode or anode?
A: In lithium-ion batteries, lithium itself is not the electrode material—lithium ions shuttle between electrodes. The anode (negative during discharge) is typically graphite (where lithium ions intercalate), and the cathode (positive during discharge) is a lithium metal oxide (e.g., LiCoO₂, NMC, LFP). Pure lithium metal is used as the anode in some experimental or lithium-metal batteries.
References:
[1]Explore electrochemical potential theory.↪
[2]Understand electrolyte and separator roles.↪
[3]Understand charge and discharge behavior.↪
[4]Explore optimized charging strategies.↪
[5]Learn why lithium plating must be avoided.↪
[6]Compare common anode materials of graphite.↪
[7]Compare common anode materials of silicon.↪
[8]See how electrode coating quality impacts battery performance.↪
[9]Learn how calendering balances porosity and energy density.↪
[10]Discover why N/P ratio optimization is critical for cycle life.↪
[11]See how thermal management improves battery safety and longevity.↪
[12]See how liquid cooling improves battery safety and lifespan.↪
[13]Explore the growing importance of battery recyclability.↪
[14]Understand how C-rate affects battery discharge performance.↪

